 
Keep in mind, however, that the VSEPR model, like any model, is a limited representation of reality the model provides no information about bond lengths or the presence of multiple bonds. , which can be used to predict the shapes of many molecules and polyatomic ions. We continue our discussion of structure and bonding by introducing the valence-shell electron-pair repulsion model A model used to predict the shapes of many molecules and polyatomic ions, based on the idea that the lowest-energy arrangement for a compound is the one in which its electron pairs are as far apart as possible. To predict whether a molecule has a dipole moment.To use the VSEPR model to predict molecular geometries.General Chemistry for EngineeringCHM 2000 Chapter : Predicting The Geometry Of Molecules Sulfur dioxide is polar in nature.The difference in electronegativity between sulfur and oxygen atoms creates polarity in the molecule.Oxygen has a greater electronegative potential than sulfur.Therefore, oxygen exerts more pull on the covalent bonds in sulfur dioxide.The portion of the molecule that has both oxygen atoms on it is slightly negatively charged.whereas the portion that has the sulfur atom has a slightly positive charge.This makes SO2 a polar molecule like H2S.In addition, the unbonded electrons on the sulfur and oxygen create repulsion between atoms.This is another cause of the polarity of the sulfur dioxide molecule.Check the full article SO2 is polar or nonpolar?. Recommended Reading: Geometry Dash Blast Processing Full Ver So2 Is Polar Or Nonpolar Lets draw the Lewis structure of SO2 using the following simple guidelines. There are a total of 5 lone pairs in the SO2 lewis structure. It is bonded with the help of two double bonds to two atoms of oxygen at the sides. The Lewis structure of SO2 consists of a sulfur atom present at the center of the molecule. So, both atoms have 6 valence electrons each. Having a quick look at the Periodic Table, we can readily identify that both Sulfur and oxygen are situated in Group VI A. Valence electrons present in an atomic element can be easily determined by using the Periodic Table. Therefore, the first step whenever we want to draw the Lewis structure of a molecule is to calculate the total valence electrons present in it. The Lewis dot structure of a molecule is a simplified representation of all the valence electrons present in the molecule. S For Drawing The Lewis Dot Structure Of So2ġ. 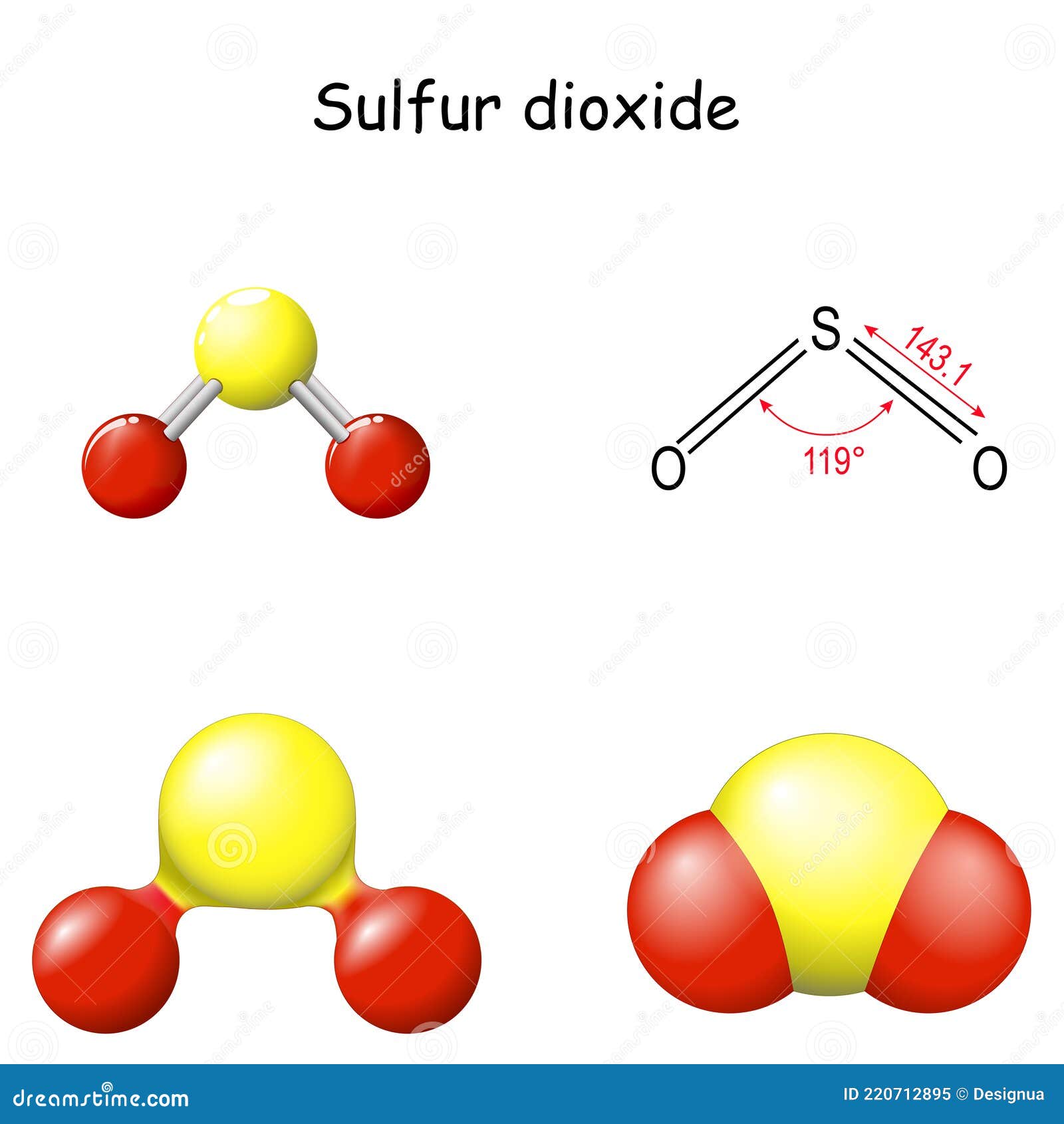
Sulfur dioxide is naturally found in small amounts in the atmosphere and is a primary precursor of sulfuric acid. It is naturally released by volcanic activity.It gives a weak acid solution when dissolved in water. It is a colorless, toxic, inorganic gas with a pungent smell like nitric acid. Sulfur Dioxide, also known as Sulfur Dioxide, is the entity of a bond between sulfur and oxygen atoms. SO2 Molecular Geometry / Shape and Bond Angles (Sulfur Dioxide) 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
